Blue dadadi
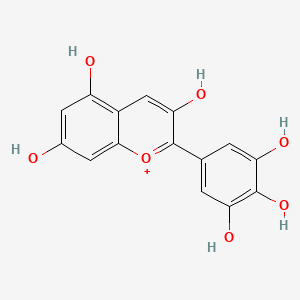
The color blue is familiar to most people and is, in fact, most peoples favorite color (2). But it may be surprising to some that blue is actually almost never found organically in nature. The blue found in animals is mostly caused by reflection of blue light by tiny structures called schemochromes, which are structural differences that cause light to interact with the structures in a certain way, causing them to appear a certain color (3). This is most apparent with blue birds such as blue jays or hyacinthine macaws. These feathers of these birds have air pockets within the bard that act as tiny particles which scatter light refracted by the oxygen in the air (3). Some plants can produce a blue pigment in this way as well but most of the blue found in plants is due to a class of chemical compounds called anthocyanins. These compounds are responsible for many pigments in nature including reds and purples, the anthocyanin responsible for the blue pigment is called delphinidin (4).
This lack of blue natural pigment is actually somewhat of a problem in the food industry, having natural, consumer safe and vibrant pigments in all colors is something food and beverage companies search for. Colorants are typically used to replace pigment lost by packaging and storage, or adding color to otherwise colorless products such as soda or frosting (1)
1 .Newsome, Andrew G., et al. "Nature's Palette: The Search for Natural Blue Colorants." Journal of Agricultural and Food Chemistry, vol. 62, no. 28, n.d., pp. 6498-6511. EBSCOhost, ezproxy.shsu.edu/login?url=http://search.ebscohost.com/login.aspx?direct=true&db=edswsc&AN=000339226700002&site=eds-live&scope=site. Accessed. November 16, 2017
2 . https://www.livescience.com/34105-favorite-colors.html Accessed. November 16, 2017
3. http://scienceblogs.com/grrlscientist/2007/10/16/schemochromes-the-physics-of-s/ Accessed. November 16, 2017
4. http://www.ddwcolor.com/natural-colours/anthocyanins/ Accessed. November 16, 2017
5. Photo credit: https://pubchem.ncbi.nlm.nih.gov/compound/Delphinidin#section=Top Accessed November 16, 2017
I actually knew a little about this from one of my classes, but your blog was really informative. It is so weird to me that blue is not commonly found organically in nature. Great post, you really helped answer questions that I had from when I heard this in lecture!
ReplyDeleteThis is very interesting because I love blue! I had no idea that is was not really found in nature, I would actually think that is it. It was even better to know the reasons why is not commonly found. Great post, I learned a lot!
ReplyDeleteI never knew that blue wasn't found in nature! It's crazy because almost everywhere you look, there is some shade of blue. I wonder if this is why blueberries are actually blue. This was super interesting post about something I never knew. Great post!
ReplyDeleteI think I had heard of this in my zoology class. It amazed me then and it still amazes me now. My mind cant comprehend how something we see inst really there. I think its weird that other colors exist organically in nature but blue doesn't. It doesn't really make sense to me. The way you explained it clears that up a bit. Nature will never cease to amaze me!
ReplyDeleteWhat does π → π* mean? n → π*
ReplyDeleteBoth represent energy transitions, when a photon of light is absorbed by a molecule electrons are promoted from low to high energy state. "Pi" in this sense is used to represent double bonds in a molecule and "n" represents non-bonding orbitals. Pi* is the higher energy anti bonding orbital and Pi is the lower energy bonding orbital.
DeleteThese two particular types of transitions are relevant in this article because the photons of light that promote these transitions are within the UV and visible light range.
Hope that answers your question somewhat.
Thanks for the clarification.
DeleteI worked with blue jays for a year and I actually never knew about this! I really never imagined it was so complicated. Overall, this is a very informative post and you do a great job of explaining why we see blue colors in nature.
ReplyDeleteThis is so interesting! I fall into the "blue is my favorite color" pile too. I had no idea about any of this. This post was very informative and helpful for someone like me that has no prior knowledge about this. I would've never thought a blue pigment would be so difficult to reproduce simple because we are so used to seeing it everywhere.
ReplyDelete